Laytons ETL | MHRA
Our regulatory lawyers have extensive experience advising businesses and individuals regulated by the Medicines and Healthcare products Regulatory Agency (MHRA) on how best to navigate regulatory challenges (see here).
If you have encountered, or otherwise wish to prepare for, a regulatory difficulty, please contact our team of regulatory lawyers for a confidential and no-obligation initial meeting.
Activity:
On 21 and 22 April 2026 we attended the Making & Distributing Pharmaceuticals 2026 conferences to support the exhibition of the Paradigm Shift Group Ltd, and to meet with other clients and experts in the industry.
Further to Ben Thorogood presenting at the 2025 conference on ‘A litigator’s perspective: when GDP goes wrong’, on 16 and 17 June 2026, we will be attending UK GDP Association Conference 2026, to continue to support CIC’s important activities in the pharma industry.
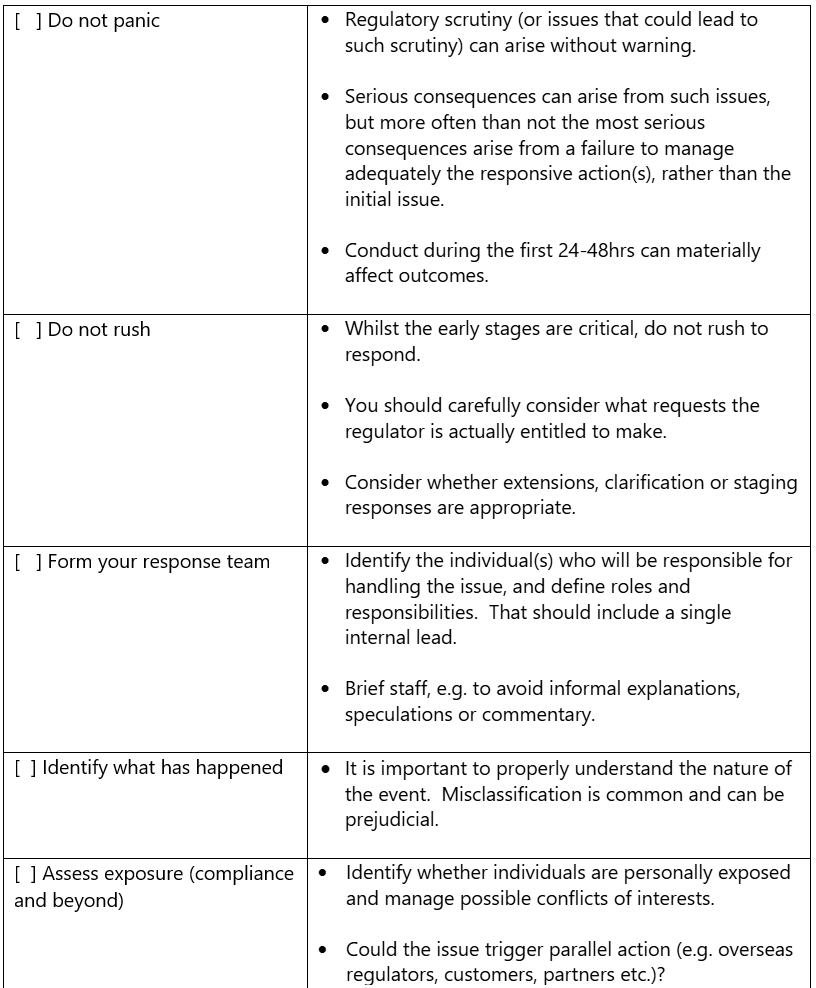
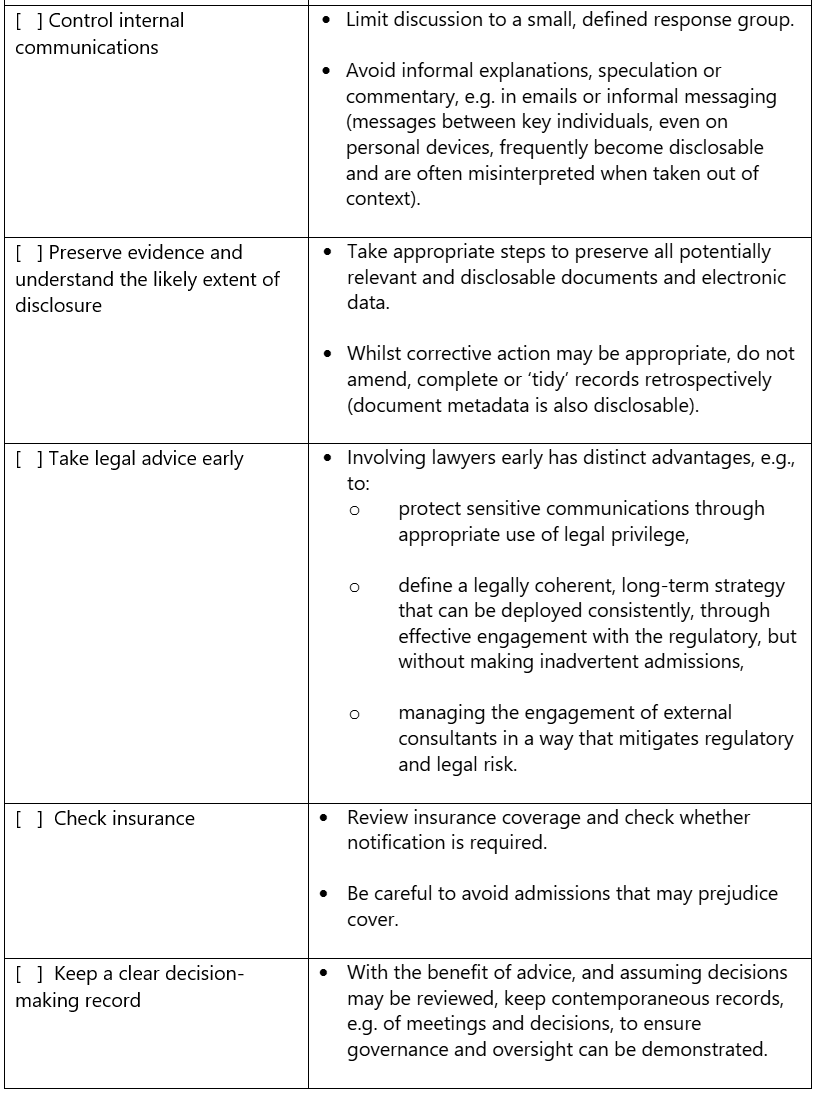
Regulatory Difficulty? - Checklist
This checklist is intended to provide businesses and individuals regulated by the MHRA with a practical guide to handing their initial response to a regulatory difficulty.
Please see a PDF version of this table here.
Important note: this checklist provides general guidance only and does not constitute legal advice. Each matter is fact-specific and the best response will depend on the particular circumstances.



Partner
Disputes | LinkedIn